Green Algae Oligosaccharides:
From Natural Bioactivity to Precision-Engineered Performance
As discussed in our previous article (https://news.agropages.com/News/NewsDetail---56562.htm), green algae oligosaccharides exhibit dual mechanisms of immune induction and antimicrobial activity, demonstrating significant potential in the field of sustainable crop protection.
To fully unlock this potential, Seawin Biotech has established a targeted derivatization technology platform to precisely modify key functional sites of green algae oligosaccharides. This article systematically analyzes modification strategies focusing on active sites such as hydroxyl and carboxyl groups, and reveals how molecular engineering enhances bioactivity, improves stability, and enables new functional properties. These advancements provide a molecular blueprint for the development of next-generation green crop protection products and integrated crop health management solutions.
The functional enhancement of green algae oligosaccharides is structurally driven. Their activity improvement relies on the presence of abundant functional groups (Figure 1), with the primary modification targets including:
1. Hydroxyl groups (-OH): The C2/C3/C4 hydroxyl groups of monosaccharide units such as rhamnose, which can undergo nucleophilic substitution or esterification reactions;
2. Carboxyl groups (-COOH): The C6 carboxyl groups of uronic acid units such as glucuronic acid, which can be subjected to ionization or amidation;
3. Reducing-end aldehyde groups (-CHO): Which can be linked to functional moieties through Schiff base reactions.
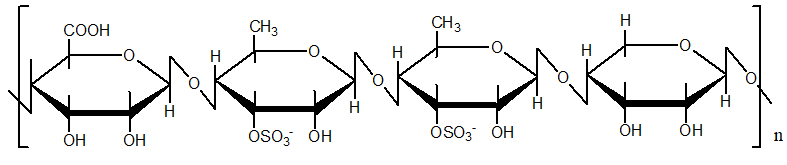
Figure 1. Modification sites of Ulva-derived rhamno-oligosaccharides (Rha-Oligo)
Depending on the desired biological activity and mechanism of action, different modification strategies can be applied to green algae oligosaccharides:
1. Among derivatization strategies, sulfation represents an effective approach to strengthen immune signal transduction.
Sulfation modification (-OSO₃H) enhances receptor affinity and increases signaling intensity, thereby amplifying the immune-inducing activity of green algae oligosaccharides.
Target site: Hydroxyl groups at the C2/C3 positions of rhamnose units
Enhancement mechanism:
(1) Sulfate groups mimic pathogen-associated molecular pattern (PAMP) structures, increasing the binding constant with receptors (such as LYK5);
(2) Activation of the salicylic acid (SA) pathway with elevated expression of the key regulatory gene NPR1.
2. To further enhance signaling synergy and improve formulation performance, phenolic acid grafting is introduced. This modification strengthens signal intensity while imparting amphiphilic properties to green algae oligosaccharides.
Target sites: Reducing-end aldehyde group + hydroxyl groups
Enhancement mechanism:
(1) Ferulic acid is linked via a Schiff base reaction to form a sugar–phenolic acid conjugate;
(2) Synergistic activation of both the SA and JA pathways, with simultaneous upregulation of PR1 and PDF1.2 genes.
Formulation advantage:
The grafted product exhibits increased lipophilicity, extending leaf surface retention time to 2.3 times that of native oligosaccharides.
3. For direct enhancement of antimicrobial activity, cationization through quaternary ammonium modification provides a structurally precise solution.
Quaternary ammonium modification (-N⁺R₃) strengthens the direct bacteriostatic activity of green algae oligosaccharides through precise regulation of charge and spatial configuration.
Target site: Quaternization following amidation of the carboxyl group
Enhancement mechanism:
Introduction of positive charges (+15.2 mV Zeta potential) disrupts the bacterial membrane potential.
Structural design:
Quaternized green algae oligosaccharides can insert into the lipid bilayer (Figure 2), increasing intracellular ATP leakage.
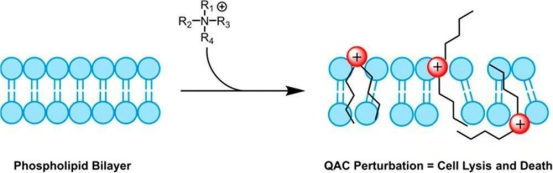
Figure 2. Schematic illustration of quaternized green algae oligosaccharides inserting into the lipid bilayer.
4. Single-site modification enhances performance, while dual-site design redefines it.
Through synergistic dual-activity engineering, molecular design achieves a biological effect of 1+1 > 2.
Application example: Sulfated–quaternized dual-modified oligosaccharides
Structural design: Sulfation at the C3 position of rhamno-oligosaccharides + trimethylammonium propyl substitution at the C2 position (Figure 3)
Mechanism of action: The introduced positive charges enhance leaf surface adhesion, the sulfate groups activate the plant immune system, and the quaternary ammonium groups directly disrupt pathogen cell membranes.
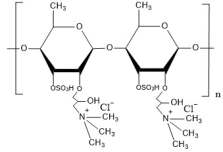
Figure 3. Structural schematic of sulfated–quaternized dual-modified oligosaccharides.
Based on the above strategies, Seawin Biotech has developed an intelligent derivatization platform for green algae oligosaccharides. This platform integrates molecular design with scalable manufacturing, ensuring that laboratory innovation can be translated into industrial feasibility:
(1) Computer-aided design: Predicting receptor binding energies through molecular docking analysis.
(2) Modular synthesis: Enzymatic targeted cleavage of oligosaccharide fragments (fixed DP) → Site-specific chemical modification (controllable degree of substitution) → Membrane separation and purification (retention rate >95%).
Through precise modification of key functional sites, green algae oligosaccharides evolve from natural bioactive substances into designable intelligent functional molecules.
l Sulfation enhances immune signal transduction.
l Cationization directly disrupts pathogens.
l Amphiphilic design improves spray performance, enhances leaf surface spreading, and delivers superior foliar application results.
Together, these advancements position green algae oligosaccharide derivatives as key enabling technologies in next-generation crop health management systems, accelerating the transition toward high-efficiency, sustainable agricultural protection.




